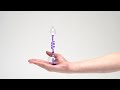
Hand Boiler
Price:
€12.78
-
Code:138-1
Hold the bottom of the flask in the palm of your hand and wrap your fingers around it. The warmth of your hand raises the temperature of the gas. The increase in temperature creates an increase in air pressure - the gas expands. This increased pressure starts to push the liquid up the glass tube to the top of the flask. When all the liquid has been moved upwards and there is only gas in the lower part of the flask, air bubbles are pushed out of the tube and it seems as if the liquid in the upper part has started to boil.
Height: 17см.